https://doi.org/10.1021/acsomega.0c03749
“To understand the mode of CO2 adsorption on zeolite ZK-4, we monitored the adsorption of CO2 on the selected zeolite ZK-4s using in situ infrared (IR) spectroscopy. Bands for both physisorbed and chemisorbed CO2 were detected in the IR spectra in Figure 5. The relative fraction of chemisorbed CO2 was higher on zeolite Na-ZK-4 (1.8) at low CO2 pressures than that at high pressures. However, irrespectively of the pressure, the major mode of CO2 adsorption was via physisorption, as derived from the very large intensity of the asymmetric stretching υ3 band at around 2360 cm–1 (the band that appeared at 2289 cm–1 was related to 13CO2). (45) The frequency of this band downshifted at increased loading of CO2, which was consistent with that CO2 adsorbed at high-energy sites at low loading of CO2. (23,46) At very low CO2 coverage, the physisorbed CO2 occurred on the high-energy physisorption sites that are typically located close to the cations. CO2 can interact with one or more cations if the cations are located close to each other there. We have previously investigated the effect of the cation on CO2 physisorption by density functional theory (DFT) calculations for zeolite A and zeolite Na-ZK-4 (1.3). We observed that at low CO2 coverage, different cations and their crystallographic sites can affect the orientation of the physisorbed CO2. As zeolite Na-ZK-4 (1.8) shares the same structure as zeolite A and zeolite Na-ZK-4 (1.3), we expect a similar behavior on CO2 adsorbed on zeolite Na-ZK-4 (1.8). (23,26) Also, we have shown by in situ neutron diffraction studies that most of the CO2 do indeed physisorb by bridging two cations in zeolite A. (24) In our previous studies, we also demonstrated using DFT calculations that the frequency of the υ3 band can be shifted depending on the cation composition near the physisorbed CO2. The high-frequency bands at 3599 and 3715 cm–1 were attributed to the combination of frequencies (υ1 + υ3 and υ1 + 2υ2) of physisorbed CO2. The normally forbidden band for CO2 (symmetric stretching υ1 band at 1383 cm–1) appeared at high loadings of CO2 and informed on that sufficiently many CO2 molecules had a distorted symmetry.”
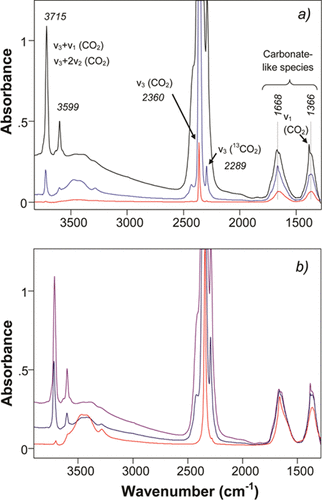
“Figure 5. IR spectra of CO2 adsorbed on zeolite Na-ZK-4 (Si/Al ∼1.8:1) in (a) adsorption branch at CO2 pressures of 6.7 Pa, 133 Pa, and 13.3 kPa (bottom to top) and (b) desorption branch at CO2 pressures of 13.3 kPa, 667 Pa, and 20 min of evacuation under dynamic vacuum conditions.”
“Although physisorption of CO2 was dominant, chemisorption of CO2 was still detectable and could be seen from the broad bands at 1668 and 1366 cm–1 (Figures 5 and 6), which were somewhat similar to the bands observed for chemisorption on zeolite A. (47−50) These bands, according to the split (∼300 cm–1) of double-degenerated carbonate bands, were assigned to bidentate carbonates. (48) As it is always very difficult to remove all the water from aluminum-rich zeolites, it is very well possible that some bicarbonates had formed upon CO2 chemisorption. (36) The appearance of the broad OH bands at 3400 cm–1 on the spectra in Figure 5 can be a further indication of bicarbonate formation. The physisorbed CO2 could be removed from the pores of zeolite Na-ZK-4 by evacuation, but the chemisorbed species remain in the zeolite (cf. Figure 6a). The fraction of chemisorbed CO2 was the highest on zeolite Na-A and then successively lower on zeolites Na-ZK-4 (Si/Al ∼ 1.8) and Na-ZK-4 (Si/Al ∼ 2.8) (cf. Figures 5 and 6a), which was assigned to the decrease in the number of Al atoms and the metal cations that provided the basicity of the zeolites. The intensities of the chemisorption bands were lower compared with studies of zeolite A. (20,23,26) Because the amount of zeolites used to make the self-supporting pellets was roughly the same (∼25 mg), the pellets had very similar thickness, and hence, a semiquantitative comparison was possible. It is important to note that apart from the number of Al atoms, the cations could also affect the chemisorption of CO2 on these zeolites. K+ is slightly less-electronegative than Na+ (0.9 vs 0.8), and the K+-containing zeolite ZK-4 would have higher basicity as compared with the pure Na+ zeolite ZK-4. Figure 6b demonstrates clearly the effect on the chemisorption of CO2 when introducing K+ on Na-ZK-4 (Si/Al ∼ 2.8). An increased intensity of the corresponding bands and shifts in the frequencies are observed in Figure 6b when comparing spectra (i) and (iii), displaying the increased degree of chemisorption of CO2 when 55 at. % of the Na+ was replaced with K+ on Na-ZK-4 (Si/Al ∼ 2.8). The split of some carbonate (or bicarbonate) bands also increased in magnitude from ∼300 cm–1 on Na-ZK-4 (Si/Al ∼ 2.8) to around 350 cm–1 on NaK-ZK-4 (Si/Al ∼ 2.8) 55 at. % K+. Similar increases in the magnitude of the split were observed when comparing Na-ZK-4 (Si/Al ∼ 1.8) and NaK-ZK-4 (Si/Al ∼ 1.8) 28 at. % K+; however, no increased amount of chemisorbed CO2 was observed on NaK-ZK-4 (Si/Al ∼ 1.8) 28 at.% K+.”

“Figure 6. (a) IR spectra of (i) zeolites Na-ZK-4 (Si/Al = 2.8:1), (ii) Na-ZK-4 (Si/Al = 1.8:1), and (iii) NaA. The bands were assigned to carbonate and/or carbonate-like species, and there are more different kinds of such species in the case of zeolite 4A and (b) IR spectra of (i) zeolites Na-ZK-4 (Si/Al = 2.8:1), (ii) Na-ZK-4 (Si/Al = 1.8:1), (iii) NaK-ZK-4 (Si/Al = 2.8:1) 55 at. % K+, and (iv) NaK-ZK-4 (1.8) 28 at. % K+.”
