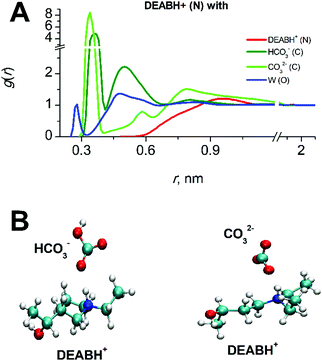
“Fig. 5 shows the molecular diffusion coefficients of all species present in solution after covalent CO2 binding to MEA. The diffusion coefficients of all species significantly decrease with increasing CO2-loading (numerical data can be found in the ESI,† Table S4). It has to be noted that the diffusion coefficient of CO2 in the fully reacted system (at 0.5 loading) is two times smaller than in the pre-reaction state since all MEA molecules have reacted with CO2. As expected, the mobility is critically dependent on the molecular mass: the heaviest molecules MEACOO− and MEAH+ are least mobile whereas water and CO2 diffuse fastest. The significant decrease of molecular diffusion coefficients is consistent with the experimentally measured increase of viscosity of the mixed solution (see above). Since hydrogen bond formation of water molecules does not change with CO2-loading (data not shown), the increase of viscosity of CO2-loaded MEA solution can only be explained by an increase of ion pair formation of MEACOO− and MEAH+ with higher supramolecular masses and lower diffusion coefficients.
 |
||
| Fig. 5 Diffusion coefficients of molecular components as a function of CO2 loading in aqueous MEA/CO2 solution. | ||
Compared to the primary amine MEA, the tertiary amine DEAB displays a different behavior upon an increase of CO2 loading. In the pre-react state, DEAB molecules tend to self-aggregate and form large clusters (see a snapshot of an entire system in Fig. S2, ESI†). The CO2 molecules show a preferred localization within these hydrophobic aggregates.14,17
In contrast to MEA, absorption of CO2 in aqueous solutions by DEAB significantly affects the liquid structure. The effect of increasing CO2 loading on DEAB cluster formation can be seen in Fig. 6A. The peak amplitudes of the first and the second peaks of the RDF characterize the tendency of DEAB molecules to form self-aggregates. At low CO2-loading, DEAB clustering even increases since the presence of only a small amount of charged particles DEABH+/HCO3−/CO32− in solution enhances the hydrophobic/hydrophilic phase separation. Upon further increase of CO2-loading, more DEAB molecules have participated in the chemical reaction with CO2 which then leads to the formation of an increasing number of protonated DEABH+ molecules. Those tend to separate from the DEAB clusters due to their hydrophilic character and the number of hydrophobic DEAB–DEAB interactions decreases. This can be seen by the decrease of peak amplitudes in Fig. 6A at high loading. At a CO2-loading of 0.815, the second solvation shell of DEAB–DEAB aggregates almost vanishes indicating the formation of isolated molecular clusters.
“