https://doi.org/10.21595/vp.2022.22497
“Due to diverse applications of Lithium Nickel Manganese Cobalt (LiNixCoyMnz, NCM) composites in advanced electronics and material/alloy, the analysis of impurity composition is an important areas for evaluating its quality. In this paper, the sulfur determination in NCM composites by Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) was proposed. The coexisting impurities effect of Si, Fe, Mn, Mg, Ca, Ni, Cr and main matrix was investigated. Using the optimized conditions, a good linear relationship of sulfur was exhibited in the range of 0-10 mg/L (r≥ 0. 9999), the standard addition recovery was 98.11-102.07 % and the RSD was 3.69 %, the levels of coexisting impurities lower than 5.0 % has no obvious interference on the determination of sulfur. This method could be served as a reliable determination of trace sulfur content in NCM composites.”
”
2.1. Reagents and chemicals
Analytical reagent (AR) grade reagents and solvents were applied for all experiments. The NCM samples were supplied by Grirem Advanced Materials t Co., Ltd., China. The Ultrapure water obtained by Millipore unit (Millipore, USA) was applied for the preparation of samples. Si, Fe, Mg, Ca, Na, Zn, Cu, Zr and S standard solution of 1000 mg/L was supplied by National Institute of Metrology. In the experiment, all the gases used (H2, N2, Ar and He) were of 99.999 % purity.
2.2. Sample preparation
In a typical process, 0.1000 g sample was accurately weighed and fully dissolved in nitric acid and heated at a low temperature without other preparation. The aliquot of solution was placed in ptfe griffin beaker for several hours and then simultaneously transfer into a 100 ml flask. All samples were passed through syringe filter before injection to the test system. At specified time intervals, about 5 ml was taken from solution and analysed by ICP-OES. All Samples were prepared according to the above process.
2.3. Standard stock solution
2.3.1. Sulfur standard solution A
10 mL of sulfur standard solution (1000 mg/L) was drawn into a 100 mL volumetric flask. Sulfur standard solution of 100 mg/L was prepared as stock solution A.
2.3.2. Multi-element mixed standard solution B
10 mL of Si, Fe, Mg, Ca, Na, Zn, Cu, Zr standard solution (1000 mg/L) was drawn into a 100 mL volumetric flask, respectively. A multielement mixed stock solution B was obtained, which included Si, Fe, Mg, Ca, Na, Zn, Cu, Zr standard solution of 100 mg/L.
2.4. Standard solutions
The sulfur standard solutions between 0 and 10 mg/L were prepared by diluting the stock solution A appropriately. The substances include amount of Ni, Co, Mn and Li were added into the liquid as a base. The calculated quantification of impurity content were carried to evaluate the effectiveness of the effect of impurity influence. A slope was used to explore the recovery values during the determining of samples.
2.5. Equipment
An ICP-OES (SPECTRO ARCOS) was used for the identification and analysis of sulfur in NCM samples.
2.6. Calculation
The value of the sulfur is determined by the calculation method as bellow:
�=(�-�0)⋅�×10-6�×100%,
where, � and �0 (μg/mL) stand for the content of the measured component in the sample solution and blank solution; � (mL) stands for the total volume of the solution; � (g) stands for the quality of sample.
3. Results and discussion
3.1. Selection of optimum elemental analysis wavelength
An ICP-OES (SPECTRO ARCOS) was used in this experiment. The main operating parameters are as follows: a peristaltic pump was used with a flow rate of 2 mL/min, transmitted power 1350 W, coolant gas flow rate 0.8 L/min, auxiliary gas flow rate 0.8 L/min and nebulizer gas flow rate 0.8 L/min (Table 1). According to the optimum instrument conditions, the spectral intensity of sulfur in standard series solution, blank solution and sample solution would be measured.
The interference-free model was investigated for sulfur by recording the spectra of the sample solution and potentially interfering factor near the analytical line. Considered about the interfering factor near the analytical line and high precision for detection, the wavelength 182.034 nm was selected for this study.
Table 1. Optimum operating parameters
|
Parameter
|
Value
|
Parameter
|
Value
|
|
Transmitted power
|
1350 W
|
Flow rate of sample uptake
|
2 ml/min
|
|
Flow rate of nebulizer gas
|
0.80 L/min
|
Atomizer pressure
|
2.8 bar
|
|
Flow rate of auxiliary gas
|
0.80 L/min
|
replicate readings
|
3
|
|
Flow rate of plasma gas
|
0.80 L/min
|
one read time
|
40s
|
3.2. Sample analysis
3.2.1. Linearity, LOD and LOQ
The method for sulfur analysis was validated on the basis of linearity, limit of detection (LOD), limit of quantitation (LOQ). Calibration standards of sulfur at concentration levels of 0, 1, 2, 5 and 10 mg/L were used to plot the calibration. Meantime, a good linear relationship of the proposed analytical model was obtained, as show in Fig. 1. The corresponding linear calibration curve (correlation coefficient) was 0.9999. Equation: �=38247�+40118.
Fig. 1. Representative standard calibration curve of sulfur
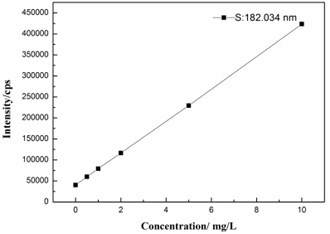
The LOD (noise ratio of 3:1) and LOQ (noise ratio of 10:1) values of this proposed method was calculated corresponding to a signal of blank samples (��), as shown in Eqs. (2) and (3), respectively:
���=3��,
���=10��.
The LOD for the sulfur was 0.0031 % while the LOQ was 0.0094 %.
3.2.2. Accuracy and precision
The average (AV) sulfur present in samples was 0.0226 %. For this, the relative standard deviation (RSD) was used to estimate the repeatability and precision. RSD was 3.69 %, which was lower than 5 % for all analytes and revealed the application of ICP-OES method for sulfur analysis (Table 2).
Table 2. Sulfur conten and precision of determination method (�= 7)
|
Sample
|
Content %
|
AV %
|
RSD %
|
|
NiCoMnLi
|
0.0221, 0.0232, 0.0228, 0.0231, 0.0212, 0.0234
|
0.0226
|
3.69
|
3.2.3. Analysis of recovery of standard addition
Standard recovery test was used to verify this method. The operation steps are as follows: weigh three parallel samples according to the same processing method above-mention. Then, added three different concentration levels of sulfur standard solution. It can be seen that the recovery rates of sulfur in NCM samples are between 91.88 % and 102.07 %. The specific recoveries of standard addition are shown in Table 3. These values are acceptable and effective for detecting sulfur in NCM samples.
Table 3. The recovery of standard addition of sulfur in NCM samples
|
Sample
|
S Concentration (%)
|
Recovery rate (%)
|
||
|
Initial
|
Add
|
Finally
|
||
|
NiCoMnLi
|
0.0226
|
0.05
|
0.0736
|
102.07
|
|
0.1
|
0.1194
|
96.83
|
||
|
0.2
|
0.206
|
91.88
|
||
”
