https://www.nature.com/articles/s41598-022-09640-9
“The role of transition metals (TMs) addition on the formation and crystallization of amorphous Al85TMs10Y5 alloys was described using in-situ high-temperature X-ray diffraction. The structural results were compared with differential scanning calorimetry and dynamical mechanical analysis to obtain detailed information about the nucleation and growth of crystalline phases. The performed analysis confirmed that Fe and Cu addition drastically changes the crystallization temperature and the phase composition of the fully crystallized alloys. While for Al85Ni10Y5 alloy, the second crystallization step is related to the formation of Al19Ni5Y3 phase, for Al85(Ni, Fe)10Y5 and Al85(Ni, Fe, Cu)10Y5 alloys crystallization of Al15Fe9Y2 phase was observed. Interestingly, the performed analysis showed that forming a homogenous amorphous phase is not necessary to obtain the best corrosion resistance. It was noted that the precipitation of the YCr2Al20 phase in the Cu-rich amorphous matrix should be a much more interesting approach.”
”
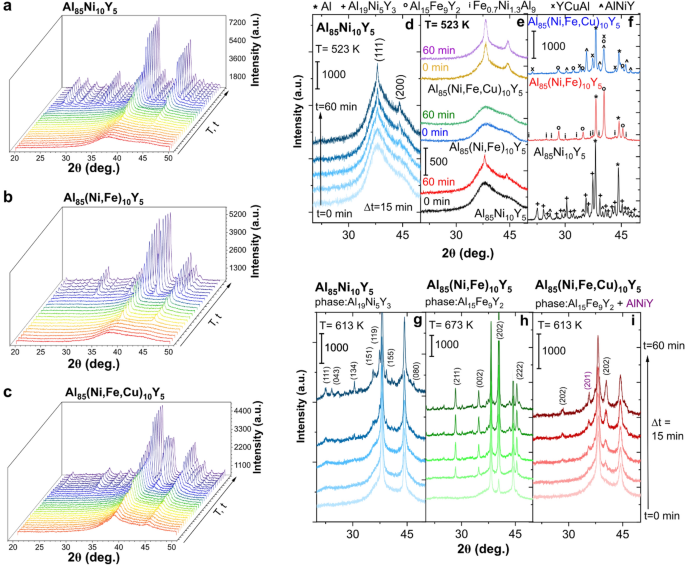
While the knowledge about the role of chemical elements in amorphous and partially crystallized alloys can be used to casting the samples with increased corrosion resistance, the information about crystallization mechanism can provide helpful information to the preparation of amorphous-nanocrystalline alloys with the controlled size of crystallites and improved mechanical properties. Crystallization from the liquid and amorphous state must be treated as two different processes. In the liquid state, the mobility of all chemical elements is very high, whereas, in the amorphous system, their movements are prolonged. Therefore, the crystallization process from an amorphous, metastable state was described for the Al85TMs10Y5 alloying system. In different studies, information about the crystallization of different alloys was determined based on DSC analysis combined with the X-ray diffraction (XRD) patterns collected for isothermal annealing samples. Sometimes, these studies are supported by analysis of TEM images. In these studies, crystallization in time and temperature domains was analyzed using the XRD method, whereas patterns were collected in-situ under an annealing sample in certain conditions. A comprehensive crystallization mechanism was determined based on recorded XRD patterns. Firstly, the evolution of XRD patterns in the time and temperature domain, where the crystallization process can be easily observed, is presented in Fig. 3a–c. As can be seen, this process is different for all analyzed alloys—the substitution of Ni by Fe and Cu influences anticorrosion properties and the crystallization from the amorphous state. However, the first crystallization step is the same and is related to the formation and growth of the Al phase. This primary crystallization results in forming a metastable structure, in which Al nanocrystals coexist with the amorphous matrix and has been observed for other Al-based amorphous alloys such as Al88Y7Fe5 and Al89La6Ni533,34. Moreover, this 1st stage can occur in the different temperature ranges for alloys containing different TMs. Analysis of crystallization of this phase in the time domain at a constant temperature equal to 523 K is presented in Fig. 3d for Al85Ni10Y5 alloy. As one can see, the intensity of two planes (111) and (200) increases simultaneously with progressive crystallization. The same situation was observed for other alloys. However, the temperature needed to start this process is different (Fig. 3e). While for Al85Ni10Y5 alloy, the crystallization of the Al phase starts at 523 K, and when the annealing time is equal to 30 min, the formation of Al crystallites cannot be observed even in the same temperature at 60 min for Al85(Ni, Fe)10Y5. On the other hand, the annealing of Al85(Ni, Fe, Cu)10Y5 amorphous alloy up to 523 K resulted in the formation of a well-crystallized Al phase. A similar finding of the role of Cu in crystallization of Al-based amorphous alloys have been presented by Kelhar et al. for Al20.5Ce41.5(FeyCu1−y)38 alloys35. The addition of Cu results in forming a fully amorphous structure in a wide Cu range (0 ≤ y ≤ 0.74), however, the Cu-rich alloys are characterized by lower crystallization temperature than alloys with higher Fe content.
In-situ XRD patterns in temperature and time-domain recorded for (a) Al85Ni10Y5, (b) Al85(Ni, Fe)10Y5, and (c) Al85(Ni, Fe, Cu)10Y5 amorphous alloys; (d) time-dependent crystallization of Al phase presented in in-situ XRD patterns of Al85Ni10Y5 amorphous alloy at a constant temperature equal to 523 K; (e) comparison of the thermal stability of amorphous phase at a constant temperature equal to 523 K for 0 and 60 min of heating; (f) phase composition of fully crystallized amorphous alloys (the markings explained above the graphics); (g) crystallization of the second (Al19Ni5Y3) phase in Al85Ni10Y5 visualized in the time domain at T = 613 K; (h) crystallization of the second (Al15Fe9Y2) phase in Al85(Ni, Fe)10Y5 visualized in the time domain at T = 673 K; (i) spontaneous crystallization of the second (Al15Fe9Y2) and third (AlNiY) phases in Al85(Ni, Fe, Cu)10Y5 visualized in the time domain at T = 613 K;
“