https://doi.org/10.1186/s40543-022-00339-0
”
ICP-OES has been established as a powerful technique for multi-element analysis. On the other hand, the technique can suffer from both spectral and non-spectral interference, limiting the accuracy, repeatability, and reproducibility of the information obtained. Because the nature of the interference is often complex, it is impossible to apply the necessary corrections for an accurate and precise analysis. Traditionally, for quantitative analysis in atomic spectroscopy, a single spectral line free from the criterion for line sensitivity and spectral interference is chosen. Many attempts have been made to correct the spectral interference of ICP-OES, including standard additions, matrix matching, inter-element correction, and optimization of the spectrum baseline selection. Nevertheless, this method has a severe limitation in calibration if the matrix of the analysis sample is complex (Griffiths et al. 2000).
The main drawback after the introduction of ICP-OES was the sample introduction system, which has a significant impact on the analysis performance of the spectrometer. The torch, one of the main areas of hardware that is coupled with the interface, is now made mostly of quartz, a crystal form of SiO2. As the plasma turns on, the temperature gradient increases along the axis. Quartz has a low coefficient of thermal expansion and is relatively resistant to rapid temperature changes, such as when plasma ignition occurs. Because of these two properties, quartz has been used for many years as an ideal material for ICP torches when designed for aqueous sample analysis. On the other hand, when an organic solvent is introduced into the plasma, the temperature gradient along the axis of the ICP torch may increase, resulting in torch devitrification and breakage. The main reason for the increase in the temperature gradient is that high radio-frequency (RF) power is generally used to analyse organic solvents. Carbon-based molecules, the main component of organic solvents, emit a large amount of infrared radiation, which is absorbed by quartz and increases the temperature of the torch (Radial Demountable Ceramic Torch for the Thermo Scientific iCAP 6000 Series ICP spectrometer 2010)
When determining Si in a sample using ICP-OES, torch devitrification causes secondary contamination of Si by SiO2, the main component of the torch, making accurate quantitative analysis difficult. For this reason, the outer tube of a ceramic D-torch, which has been recently applied for various purposes, is made of sialon, a ceramic material derived from silicon nitride, which is durable and stable at high temperatures. The Centre tube is made of alumina, which has excellent chemical and heat resistance properties. Hence, stable analysis can be performed without torch deformation, even in high-temperature plasma. In particular, a D-torch does not contain any SiO2, so there is no problem with the background derived from quartz; it is very powerful in measuring small amounts of silicon.
Many studies have been conducted to analyse impurities in petrochemical products, such as silicon, e.g. naphtha with a low specific gravity and high volatility. Methods for the quantitative analysis of more than tens of ppb have been presented through various types of pretreatment. On the other hand, most of the studies published thus far used two methods, either diluted with a less volatile solvent, such as kerosene, or the solvent is generally cooled before being introduced into the plasma using a cooled spray chamber (Asendorf 2017). These analysis methods have a high likelihood of reduced sensitivity due to the dilution of the sample. Moreover, data errors are likely to occur because the matrix of the naphtha diluted in the solvent and the calibration solvent do not match. In addition, because all of the solvents used in these methods are insoluble in water, an additional oil-based standard solution must be prepared for these experiments, and an expensive cooling device must be installed separately. In this study, propylene glycol monomethyl ether acetate (PGMEA), which is soluble in water and easily diluted in naphtha, was used as a solvent. The conventional method for Si determination was performed by dilution with a highly volatile solvent as mentioned before. The purpose of this study is to overcome the disadvantages of the dilution method to easily and accurately determine Si by trapping it in PGMEA which is non-toxic and has low volatility and high stability against plasma of ICP-OES and selectively evaporating only highly volatile naphtha.
”
”
Instrument
Thermo Scientific iCAP-7400 ICP-OES was used for silicon determination in naphtha. A ceramic D-torch and 1.0 mm alumina inner tube were used. The spray chamber used a cyclonic spray chamber made of PTFE (polytetrafluoroethylene) material that is resistant to HF and a 400 µl/min PFA (perfluoroalkoxy) microflow nebulizer at room temperature (19°C) without a separate cooling device. The hot plate used for naphtha and PGMEA evaporation was a product of Savillex (USA, HPX-200). The heating work surface area was 292 mm× 212 mm, and was used at 150±2°C. The temperature at the edge is slightly higher than the temperature outside inside the fume hood. The container used for volatilization was prepared by making PFA containing no Si with a height of 40 mm and an area of 490 mm2 and washed with HF solution at 150°C for 24 hours.
Reagents and standard
As a verification method to ensure the accuracy of the analysis results, all procedures, including pretreatment, were performed in the same manner as the certified reference material (CRM), and the analyte and results were compared. On the other hand, a reference material (RM) based on the same matrix can be used if there is no CRM. In this experiment, octanmethylcyclotetrasiloxane (OMCTS, Sigma-Aldrich, CAS No. 556-67-2, density 0.956 g/ml, boiling point 175°C), which was diluted to 200 ppb with the silicon in naphtha to prepare RM, was used. This is one of the siloxanes commonly present in naphtha and causes major silicon contamination problems.
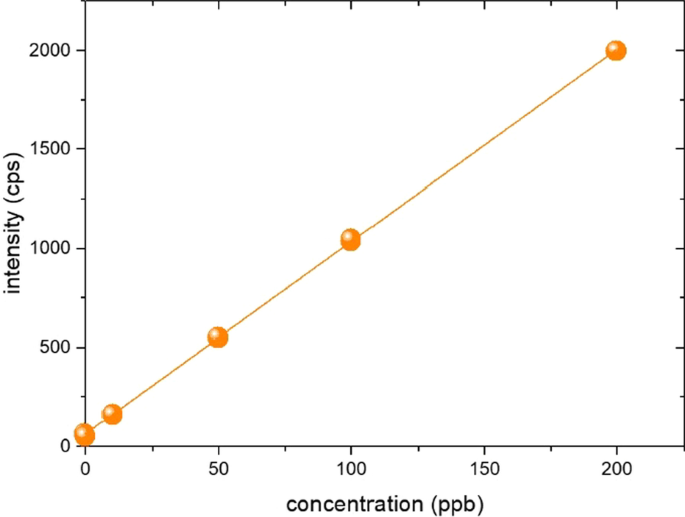
In ICP-MS analysis, the ideal matrix for the sample is water, but naphtha is insoluble in water. Therefore, a 10 ppm mother standard solution was prepared by diluting 1000 ppm Si single standard (AccuStandard, USA) in PGMEA which is soluble in water, and then re-diluted in PGMEA to 10 ppb, 50 ppb, 100 ppb, 200 ppb concentrations to prepare a standard solution for a calibration curve. RM was prepared at 200 ppb by diluting 10 ppm mother solution directly in naphtha.
Naphtha evaporation rate measurement
Naphtha used in the experiment was placed in a clean polyethylene bottle (Aicellomilim, HR013) and stored frozen at − 20°C. Subsequently, 5 g of naphtha was placed into the washed PFA beaker, and 10 g of PGMEA was added. The resulting mixture was heated to 80°C, 100°C, 110°C, 120°C, and 130°C on a hot plate. The weight change was measured at 10-minute intervals for 300 minutes, and the temperature at which all naphtha volatilized was set as the endpoint where the residual liquid became 10 g.
Sample preparation
After 5 g naphtha was placed into a cleaned PFA beaker, 10 g of PGMEA was added, and the hot plate temperature was set to 120°C for 15 g. The amount of residual solution was then recorded at 30 minutes intervals from 90 minutes until 210 minutes. Naphtha samples contaminated with approximately 50 ppb, 100 ppb, and 200 ppb Si were selected to check the accuracy and quantitative limit of the analysis result. The concentration change according to the volatilization time was checked to verify the reliability of the analysis result. Because this work aimed to quantify Si at the ppb level, the silicone concentration did not need to be higher. In addition, to check the loss of silicon components that may occur during the pretreatment process and ensure accuracy, the OMCTS RM sample prepared in 200 ppb was measured in the same manner as the naphtha sample, and 100 ppb samples spiked directly to the naphtha.
Results and discussion
Calibration
Since naphtha was diluted in PGMEA and all naphtha was volatilized on the hot plate, almost no naphtha was present in the sample for analysis. Therefore, there is no need to consider naphtha in ICP-OES optimization, and most of the final components are PGMEA as shown in Fig. 1. For the optimization of ICP-OES, after preparing PGMEA containing 100 ppb Si to maintain the same matrix as the sample for analysis, the RF power, coolant flow, nebulizer flow, auxiliary flow, and additional gas flow were tested to determine the condition with the largest signal size compared to the equipment background. In ICP-OES, silicon can be measured with two wavelengths of 288.158 nm and 251.611 nm. In this experiment, 251.611 nm with high relative sensitivity was selected (Amais et al. 2013; Gazulla et al. 2017), and the axial view mode was used for detection mode. The test was conducted by varying the RF power from 1150 W to 1500 W. At less than 1350 W, the background did not change, but the sensitivity was lowered, and the background was increased to 1350 W or higher. In the nebulizer, the plasma was unstable at 0.5 L/min or more, so the sensitivity of the equipment was lowered, and the RSD% (relative standard deviation) fluctuated. The ideal signal was obtained at an auxiliary gas flow of 0.5 L/min. If the additional O2 gas is insufficient, a large amount of carbon is formed from the unstable combustion, making the plasma unstable. If too much oxygen is supplied, the plasma is turned off. While checking the plasma light, the amount of O2 gas was determined so that the carbon emission in the plasma was invisible. Table 1 lists the instrumental parameters of the ICP-OES measurement for Si analysis.
”