https://doi.org/10.1038/s41598-022-17228-6
“Dimerization is critical for transcription factors (TFs) to bind DNA and regulate a wide variety of cellular functions; however, the molecular mechanisms remain to be completely elucidated. Here, we used high-speed atomic force microscopy (HS-AFM) to observe the dimerization process for a photoresponsive TF Photozipper (PZ), which consists of light–oxygen–voltage-sensing (LOV) and basic-region-leucine-zipper (bZIP) domains. HS-AFM visualized not only the oligomeric states of PZ molecules forming monomers and dimers under controlled dark–light conditions but also the domain structures within each molecule. Successive AFM movies captured the dimerization process for an individual PZ molecule and the monomer–dimer reversible transition during dark–light cycling. Detailed AFM images of domain structures in PZ molecules demonstrated that the bZIP domain entangled under dark conditions was loosened owing to light illumination and fluctuated around the LOV domain. These observations revealed the role of the bZIP domain in the dimerization processes of a TF.”
”
HS-AFM observations of PZ molecules
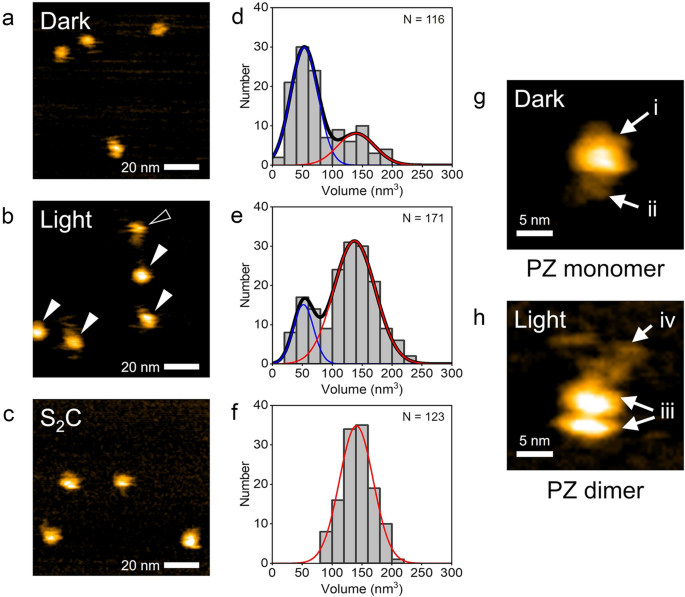
We observed the purified PZ molecules using HS-AFM under dark and light conditions in the buffer solution described in the methods section. Figure 2a shows a still image from a successive HS-AFM movie for wild-type PZ captured under dark conditions (Supplementary Movie 1). Globular-shaped molecules ~ 5 nm in diameter were visualized in the AFM image. They were well dispersed and moved rapidly on the mica surface (see also Supplementary Movie 1. The diffusing motions of the molecules will be further discussed in Fig. 3). Occasionally, molecules ~ 10 nm in diameter were also observed (see Supplementary Fig. 2). Figure 2b shows a still image of the HS-AFM movie for wild-type PZ captured under continuous BL illumination (Supplementary Movie 2). Globular-shaped molecules with two diameters were observed in the AFM image. The smaller ones were ~ 5 nm in diameter, and the larger ones were ~ 10 nm. The smaller ones rapidly diffused on the mica surface, whereas the larger ones slowly diffused (see also Supplementary Movie 2). In this HS-AFM movie, a tail-like structure was observed to fluctuate around a globular structure in the larger molecules. To compare these molecular structures with the stable PZ dimer, we observed dimeric mutant protein S2C using HS-AFM. Figure 2c shows a HS-AFM image of the mutant S2C molecules under light conditions. The globular molecules were observed, and they exhibited similar diameters to the larger molecules of wild-type PZ under illumination. These molecules slowly diffused on the mica surface (Supplementary Movie 3). In contrast to wild-type PZ, the smaller molecules were not observed in the HS-AFM movie of mutant S2C. We also observed S2C molecules under dark conditions and obtained similar results to that of light (Supplementary Fig. 3). To quantitatively compare these results, we analyzed the volumes of the molecules in the HS-AFM images. Figure 2d–f shows histograms for the molecular volumes analyzed from the HS-AFM images of wild-type PZ under dark and light conditions and from the images of mutant S2C. The histograms of wild-type PZ under dark conditions (Fig. 2d) and light conditions (Fig. 2e) show bimodal distributions, whereas those of mutant S2C show unimodal distributions (Fig. 2f, Supplementary Fig. 3). The mean volumes of the mutant S2C under dark and light conditions were similar to that of the major distribution with higher volumes of wild-type PZ under light conditions. In addition, the mean volume of the major distribution of wild-type PZ under dark conditions was similar to that of the minor distribution with lower volumes of wild-type PZ under light conditions. Therefore, we conclude that the major molecules observed under dark conditions are PZ monomers and that the larger molecules observed under light conditions are PZ dimers. Here, we note that the minor distribution of the larger molecules is seen in Fig. 2d. These molecules are dimers under dark conditions. The results of volume analyses also show that PZ predominantly exists as a monomer under dark conditions and as a dimer under light conditions. These results are consistent with a previous study involving SEC18, which revealed oligomeric states of PZ under each condition. Thus, we demonstrated that PZ monomers and dimers can be distinguished by HS-AFM. The mean volume ratio of the monomer to dimer was around 1:2.6, which differs slightly from the expected ratio of 1:2 due to dimerization. We could not clarify the major cause of this difference, but the fast diffusion of the monomer might have affected its molecular volume analysis. However, our conclusion that monomers and dimers can be distinguished by HS-AFM is not affected.
HS-AFM observations of PZ molecules. (a, b) HS-AFM images of PZ molecules observed under dark and light conditions, respectively. An open arrowhead indicates the smaller molecule with sizes of ~ 5 nm and filled arrowheads indicate larger ones of ~ 10 nm. (c) HS-AFM image of mutant protein PZ-S2C observed under light conditions. These show still images of successive AFM movie. See also Supplementary Movies 1, 2 and 3. Several PZ and PZ-S2C molecules were observed on mica surface. (d–f) Histograms for the molecular volumes analyzed from AFM images of wild-type PZ under dark, light condition and S2C mutant. N indicates the number of different molecules analyzed for each histogram. Curve represents the fit to a Gaussian distribution. Mean value of each Gaussian curve: (d) 1st peak = 52.9 nm3, 2nd peak = 139 nm3, (e) 1st peak = 52.4 nm3, 2nd peak = 137 nm3, (f) 141 nm3. (g) Highly magnified AFM image of PZ monomer under dark conditions. This molecule was included in the major distribution in (d). The folded-string structure was seen around the spherical structure. (h) Highly magnified AFM image of PZ dimer under light conditions. This molecule was included in the major distribution in (e). The dimer exhibited a bilobed structure together with the tail-like structure. Protein concentration: (a) 25 nM, (b) 50 nM, (c) 20 nM. Scan ranges: (a–c) 100 × 100 nm2, (g, h) 24.5 × 24.5 nm2. Scan rates: (a, g) 1 s/frame, (b, c and h) 0.5 s/frame. Height scales: (a) 2.5 nm, (b) 3.5 nm, (c) 4.3 nm, (g) 2.1 nm, (h) 2.5 nm.
“